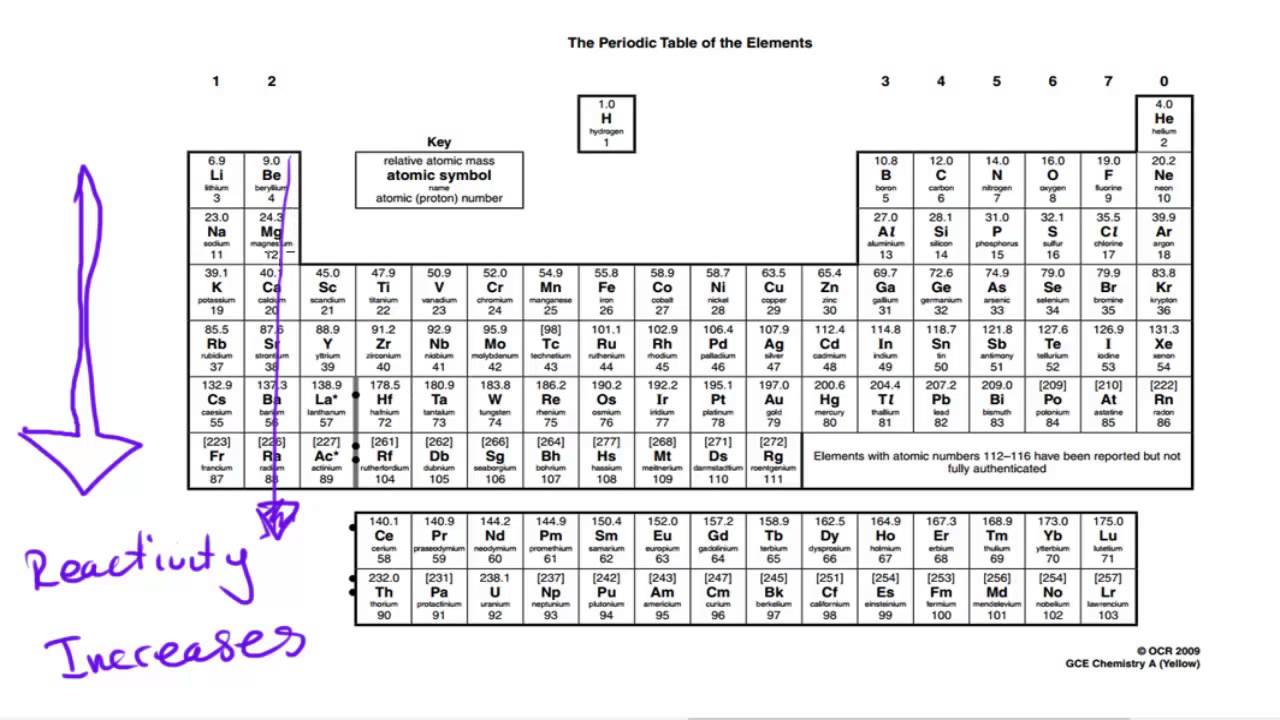 
To remember how the reactivity of the alkali metals and halogens increases or decreases, put a pin in the middle of the periodic table and spin it anti-clockwise. The outer shell will more easily attract another electron, which needs an electron to complete its full outer shell, when there is more attractive force.Ī useful mnemonic picture to help you recall that:Īs you go up group 7 (the halogens), again the elements get more reactive. The fewer electron shells (rings) between the nucleus and the outer shell (ring) also has less shielding effect and again this increases the electron attraction. The distance "a" is less than "c" and the force of attraction between the nucleus and the outer shell increases with shorter distances. Halogens from bromide to fluorine get more reactive because the force of attraction between the nucleus (core) and the outer electron get stronger as you go up group 7 elements. Why do halogens get more reactive going upwards in group 7? The outer electron is more easily transferred to say an oxygen atom, which needs electrons to complete its full outer shell. The more electron shells (rings) between the nucleus and outer electron also creates shielding and again this weakens the nuclear attraction. The distance "c" is greater than "a" and the force of attraction between the nucleus and the outer shell (rings) diminishes with distance. Why do alkali metals get more reactive going down group 1?Īlkali metals from lithium to potassium get more reactive because the force of attraction between the nucleus (core) and the outer electron gets weaker as you go down group 1 elements. This causes the electrons to experience a greater force of attraction as you move along the period, making it easier for an atoms to gain more electrons to become ions.As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive.Īs you go up group 7 (the halogens), again, the elements become more reactive. The charge on the atomic nucleus increases as you move go across the period but the electron shielding caused by the two inner electrons remains the same.The reactivity increases as you go across the period because: Nitrogen, Oxygen and Fluorine can all gain electrons to become negative ions in certain reactions. This causes the electrons to experience a greater force of attraction as you move along the period, making it harder for the atoms to lose electrons and become ions. The outer electrons are all roughly the same distance away from the nucleus.Some elements are unreactive and need energy putting in others will react spontaneously and easily. All the elements in Group 2 have two electrons in their valence shells, giving them. Reactivity of Elements of the Periodic Table Reactivity is a measure of how easily an element will combine with other elements to form compounds. Although many characteristics are common throughout the group, the heavier metals such as Ca, Sr, Ba, and Ra are almost as reactive as the Group 1 Alkali Metals. 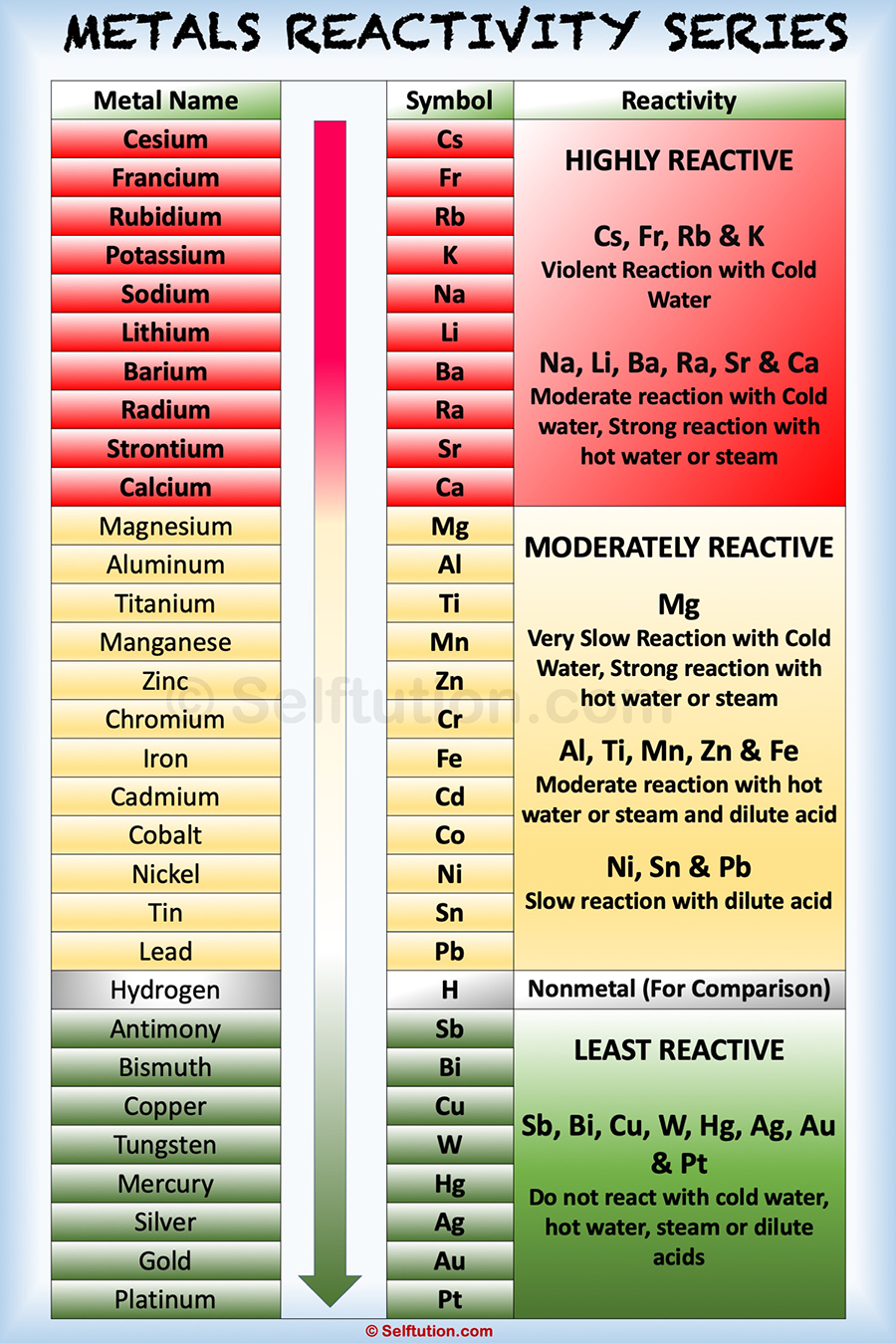
The reactivity decreases as you go across the period because: Group 2 contains soft, silver metals that are less metallic in character than the Group 1 elements. For the first 3 elements Lithium, Beryllium and Boron all lose electrons in chemical reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
